Table of Contents
You will read in this tutorial about band theory of solids. Different types of bands present in solids like valence band, Conduction band and Forbidden gap which is also known as energy band gap.
Also you will know about different roles of electrons, like valence electron, free electron etc.
ENERGY BAND
First of all, what is a band? It is a collection of energy levels.
BAND THEORY OF SOLIDS
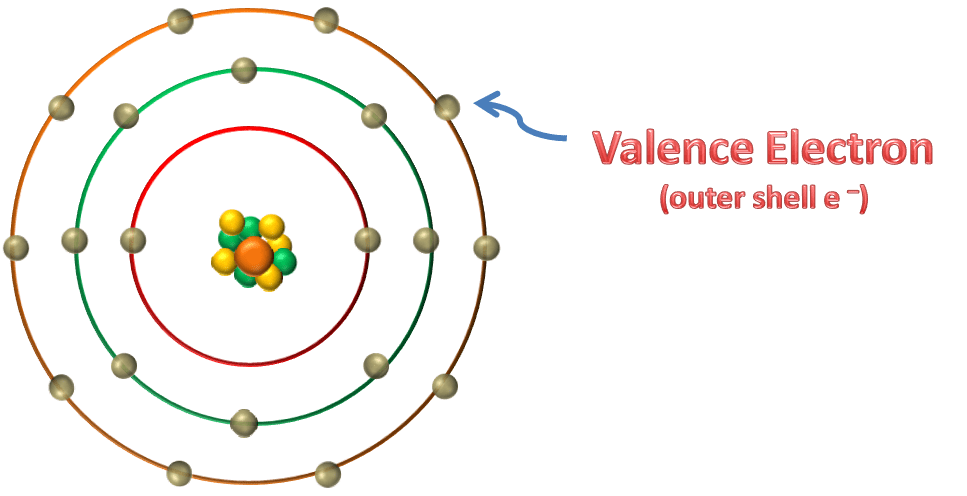
VALENCE BAND AND VALENCE ELECTRON
Electrons in outer shell are called valence electrons. In an element there are many atoms present and the valence electrons of these atoms have different energies associated with them because
Electrons of the atoms also get affected from the nucleus or electrons (e–) of neighbouring atoms (attractive force from neighbouring nucleus), so electrons have different energy levels, for example – suppose there are two electrons present in valence shell,
- One electron of valence band means one valence electron has energy of 4 but due to some forces from the neighbour atom valence electron loses its energy from 4 to 3.6
- While second electron has energy 4 and gains energy from the neighbouring atom from 4 to 4.4
So the range of these energy levels is 3.6 to 4.4 this is called a band and this band is called as valence band.
However, the energy levels of inner orbit electrons are not much affected by the presence of neighbouring atoms because they are tightly attracted to their own nucleus.
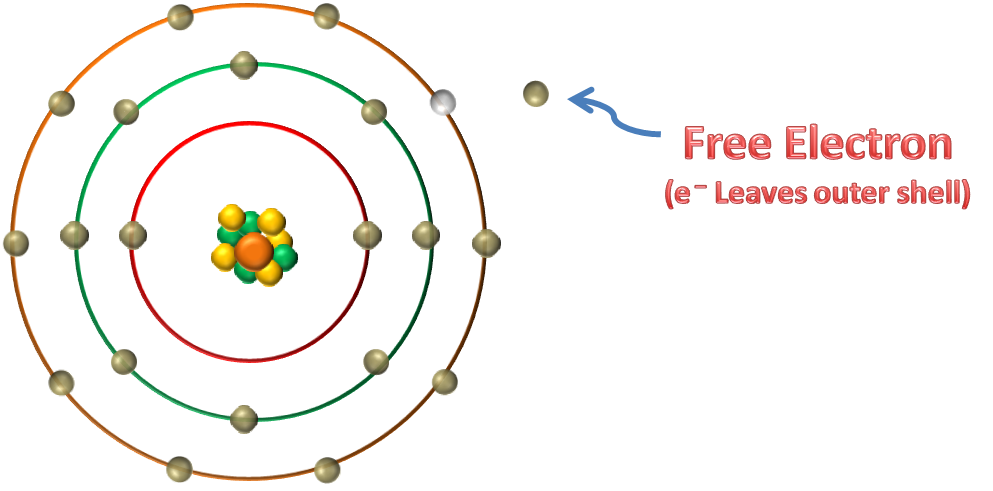
FREE ELECTRONS
Valence electrons that get enough energy to jump from valence band to conduction band are called as free electrons. In other words valence electrons that get enough energy to leave the outer shell of an atom are called as free electrons.
Valence electrons when becomes free electrons are responsible for conduction. In simple words student near the exit gate can easily go out from the class, now as they are free they can roam in the school or college premises.
These free electrons are responsible for conduction in a substance.
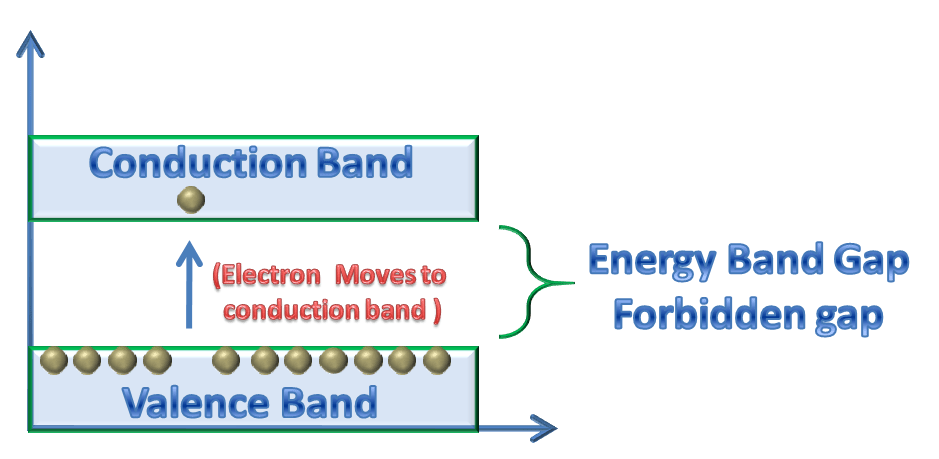
CONDUCTION BAND
Free electrons have different energies (energy levels) and the Range of energy levels of these free electrons forms a band, this band is called conduction band. Electrons in conduction band have higher energy than electrons in valence band or in forbidden band gap.
When a crystalline structure (ordered arrangement of atoms) (any object like lunch box, water bottle etc. are crystalline structure) forms, the free electrons cause current to flow through the surface of it (conductors or semiconductors) by jumping from valence band to conduction band.
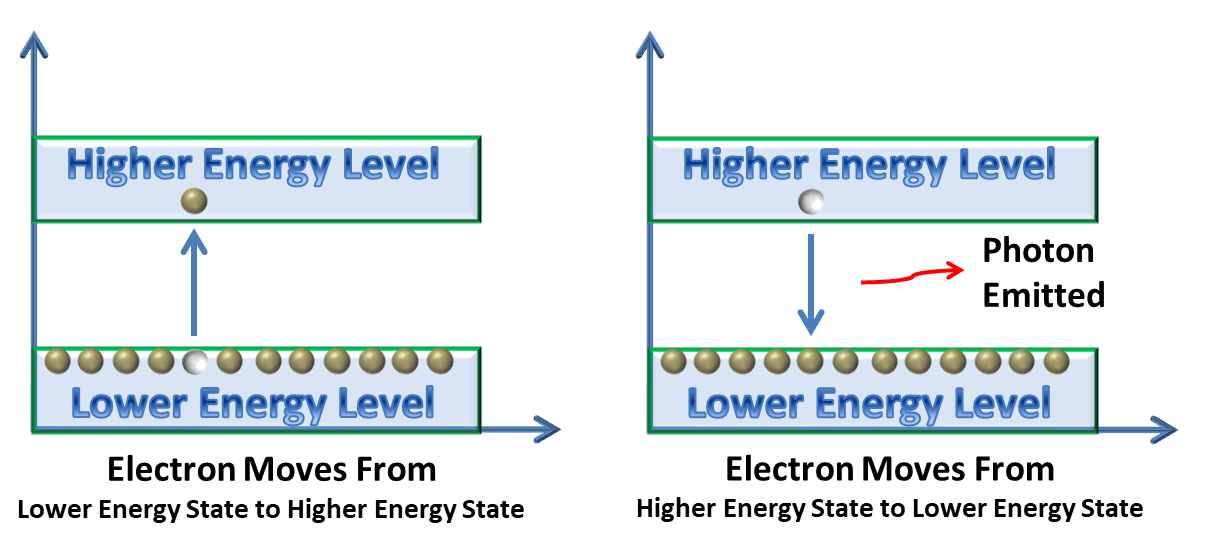
When an electron of less energy gets some more energy it jumps to upper energy level i.e. from nth shell to n+1th shell (n+1th shell have more energy than nth shell).
For conduction, a valence electron must need the energy that should be equal or greater than the forbidden energy gap to jump from valence band to conduction band.
PHOTON EMISSION
When an electron jumps from lower energy level to upper energy level it stay there for very less time and returns back to lower energy level by emitting photon (its energy). Lifetime of an electron in an excited state is a few Nanoseconds.
Now it is interesting how emitted photon can be red or green or any colour? Actually it depends on the wavelength of the emitted photon, if its wavelength lies between ranges of red colour it gives us red light or if its wavelength lies between ranges of green colour it gives us green light. So it depends on wavelength of emitted photon.
FORBIDDEN OR ENERGY BAND GAP
The energy levels difference between valence band and conduction band forms an energy band, this band is called a forbidden band gap. In other words the energy required by electrons to excite or to jump or to move from valence band to conduction band is known as Energy band gap or forbidden band gap.
NEXT POST
CLASSIFICATION OF MATERIAL BASED ON BAND GAP
PREVIOUS POST
ATOM, ORBITS AND ENERGY LEVELS